| Home A B C D E F G H I J K L M N O P Q R S T U V W X Y Z |
|
Home |
Botox Cosmetic Injections
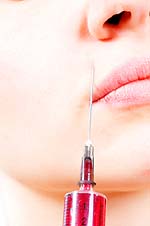
Botox Cosmetic is the number one non-surgical medical procedure for the removal of facial wrinkles, and is certainly worth examining. This article takes a close look at the science and reasons behind this relatively new cosmetic procedure. This article includes statistics and some of the unfortunate folly that has turned a viable medical procedure into a popular party drug. Contents What is Botox Cosmetic? If you could effectively erase frown lines across your brow would you do it? Thousands of people say a resounding Yes! each year, making Botox injections the number one cosmetic procedure performed. What is Botox Cosmetic? It is the brand name for Botulinum Toxin Type A. This protein complex is produced by the bacterium Clostridium botulinum, containing the same toxin that causes food poisoning. When used in a medical setting using an injectable form of purified botulinum toxin, Botox can block the release of the chemical (acetylcholine) that signals muscle contractions in the face. By selectively blocking the muscles' ability to contract, Botox can erase existing frown lines. Within just one week, frown lines are nearly invisible. According to the American Society for Aesthetic Plastic Surgery (ASAPS), Botox has become so popular, since 2001 it has surpassed breast augmentation by nearly 50% each year. In many cosmetic circles it has earned the reputation as being the ultimate fountain of youth. Botulinum Toxin Type A is not a new wonder drug. It has been around since 1983. It was approved by the FDA in 1989 to treat certain eye conditions, such as blepharospasm, where the eyelid will not stay open, and strabismus, where the eyes do not line up properly. By 2000 the toxin had also been approved to treat certain types of dystonia (a serious neurological disorder that causes severe contractions of the muscles), and hyperhidrosis (severe sweating of the armpits). Ironically in all these medical treatments, doctors observed that Botox softened the vertical frown lines between the eyebrows; lines that tended to make people look tired, angry, or old. After clinical research that showed Botox as an effective treatment in reducing the severity of frown lines for up to four months, the FDA granted its approval. In 2002. Allergan Inc. of Irvine, California began marketing Botox as the cosmetic enhancement wonder of the Twenty-First Century. For both cosmetic and neurological purposes, improvements yielded by Botox are temporary. Treatment must be repeated every 3 to 4 months. Because of this, some patients can build up immunity to the toxin, rendering the treatment ineffective. When it comes to Cosmetic Botox, however, the reverse seems to be true. The more treatments a patient has within a one-year period, the better the results and the longer the improvement lasts between each treatment. The FDA may regulate products by approving them or not, but they do not control how each product is used. Approved products are sometimes used for purposes other than those stated on the product label. Botox Cosmetic is currently being used by some practitioners to treat facial wrinkles other than those specified by the FDA. Consumers need to be aware that these off-label uses have not been independently reviewed by the agency, and may not be safe. Without proper research and studies, there is no way to determine what long-term results or risks might be involved. Ella L. Toombs, M.D., a dermatologic medical officer in the FDA's Office of Cosmetics and Colors, says Careful deliberation, investigation and evaluation are undertaken by the agency before any prescription product is approved. Drugs like Botox are subject to a greater level of scrutiny because of the benefit-to-risk ratio. In other words, the FDA may allow a greater risk ratio for patients with serious or life threatening medical conditions, as opposed to patients who are being treated for cosmetic purposes. Plastic surgery parties, seminars, and social gatherings are key elements of Botox marketing in much of the United States. A group of middle-aged men and women get together for a Botox party. They hire a doctor for the event; and split the costs of treatment. Refreshments are served, which often includes alcohol. (It should be noted that Botox injections should never be mixed with alcohol). One by one, as their name is called, each patient slips away to a private exam room. A fee is paid, and the patient signs an informed consent agreement. Anesthesia is rarely used, but sedatives and numbing agents are usually made available. The practitioner injects about one-tenth of a teaspoon of toxin into specific muscles of the forehead, most often targeted for the effect. Afterwards, the person rejoins the party. Scott A. Greenberg, M.D., a board-certified plastic surgeon in Winter Park, Florida, has been hosting monthly Botox Happy Hours in his medical office since the drug was first approved. He feels that these events are an opportunity to treat a lot of people at one time in a relaxed but professional atmosphere. He sees no difference in treating one patient or ten. According to Greenberg, identical standards of medical care are maintained at these gatherings as in a routine daytime office consultation. Julianne Clifford, Ph.D., of the FDA's Division of Vaccines and Related Products Applications, disagrees. She warns that Botox is licensed for marketing and distribution as single-use vials. In other words, each vial is intended to be used for a single patient in a single treatment session. Botox contains no preservative to protect against cross-contamination of patients. Once opened and diluted, Botox must be used within four hours. Treating multiple patients using one vial is expressly prohibited. That prohibition is stated in writing three times: on the carton, on the package insert, and on the vial itself. Franklin L. DiSpaltro, M.D., 2002 president of the ASAPS, warned that something is lost when patients are mass treated. One of my concerns, he said, is that these parties are a marketing tool. Gathering as many patients as possible trivializes a medical treatment, which could deteriorate over time into a nonprofessional environment. It seems that Dr. DiSpaltro's warning has recently become a reality. Botox parties are all the rage in many major cities. Baskets of syringes filled with Botox await party guests, who arrive for a night filled with fun and frown line-erasing injections. A technician, not a professional doctor, is available to assist party-goers with their injections. Alarming as it may be, Botox in 2006 has become one of the most popular black-market party drugs, second only to date-rape drugs and crack-cocaine. The ASAPS also reports that unqualified people are dispensing Botox in salons, gyms, hotel rooms, home-based offices, and other retail establishments. Persons who fail to seek the services of a qualified medical professional for their Botox injections run the risks of improper technique, inappropriate dosages, loss of facial control, infections due to unsanitary conditions, and disease. According to Dr. Toombs, Botox is a prescription drug that should (only) be administered by a qualified physician in an appropriate medical setting. He says that even the most skilled health-care provider can experience complications. There is no chance of contracting botulism from Botox injections; however there are other risks associated with the procedure. If too much toxin is injected or if it is injected into the wrong facial area, a person can end up with droopy eyelid muscles (ptosis) that could last for weeks. Other common side effects are headache, respiratory infection, flu syndrome, and nausea. Less frequent reactions included pain in the face, redness at the injection site, and muscle weakness. These reactions are mostly temporary but could last several months. Botox Cosmetic injections are a medical procedure that should only be administered by a qualified medical professional. Botox Cosmetic is relatively safe in the hands of a competent physician. The FDA recommends that Botox Cosmetic be injected no more frequently than once every three months, and that only the lowest effective dose needed should be administered. The prudent consumer who cares about his or her face will research a doctors credentials and do a thorough background check before agreeing to allow that physician to administer Botox injections. A qualified dermatologist, one who is board certified through the American Academy of Dermatology or the American Society for Dermatologic Surgery, would be the wisest and most reasonable place to start. Remember, it is your face. The goal is to look more youthful, without putting your health at risk. Copyright 2006: Remedium. This article may not be copied, in whole or in part, without the written consent of Remedium. |
||
|
|
||
|
Glossary References Links Contact
|